Rakshit is committed to maintain best Quality Standards in Research and Manufacturing of Pharmaceutical products through periodic reviews and continual improvement of Quality Management System, and strive to enhance customer satisfaction.

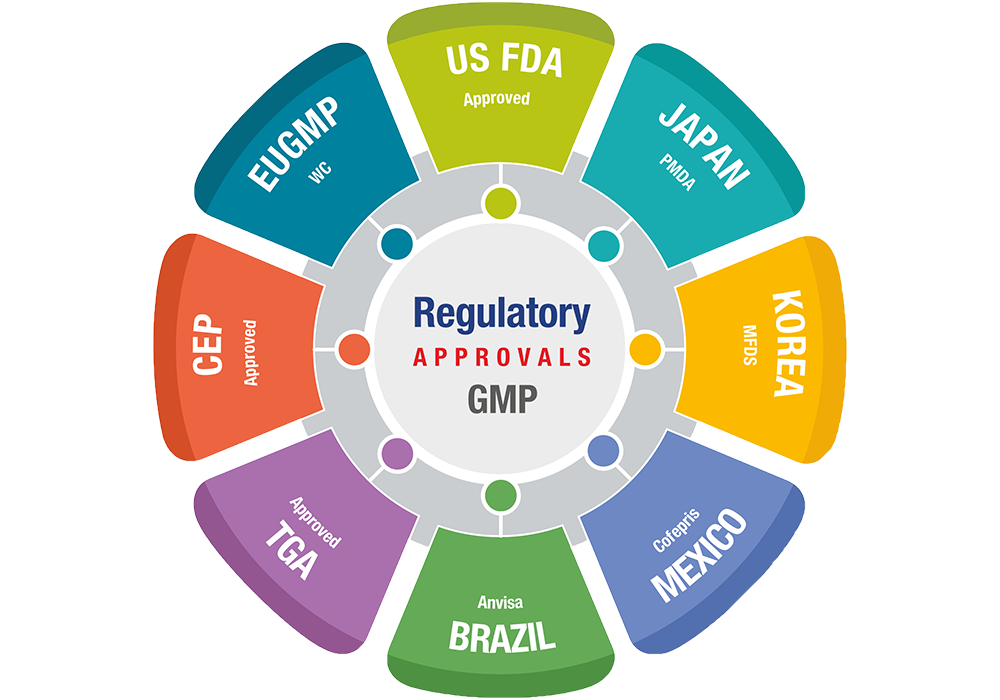
Rakshit is fully committed to ensuring our manufacturing facilities comply with international regulations for current Good Manufacturing Practice (cGMP); GLP and all locally applicable regulatory requirements.
We have been exporting to over 80 countries, with our facilities having been audited by Global Regulatory agencies like the USFDA, EDQM, EU GMP, TGA Australia, ANVISA Brazil, COFEPRIS, Japan PMDA, Korea FDA, CDSCO-India and WHO-GMP